|
From the concept of orbitals, one explains molecular structure, chemical bonding, hybridization etc and also things like spectrum of various materials. Only when the idea of electrons as particles in atoms was introduced, attempts were made to understand chemical bonding and other physical and chemical properties.Īs of now, the electron cloud model which incorporates the principles of quantum mechanics is the most successful. 
Prize motivation: for the discovery of new productive forms of atomic theory. These models were used to explain and hence to understand properties of matter. Affiliation at the time of the award: Berlin University, Berlin, Germany. The protons and neutrons form the atom’s central nucleus. It was soon realized that the model was flawed and soon Bohr put up his model in which he used Quantum concepts of Planck and Einstein to explain stability of matter and the Hydrogen spectrum.Īnd they process continued when Sommerfeld proposed his model. The rest consists of three basic types of subatomic particles : protons, neutrons, and electrons. In his model, he took the electrons embedded into the positive sphere because he found that these electrons (not some positively charged particle) were emitted by a vast range of marerials.įurther, when Lord Rutherford discovered that the positive charge and most of the mass was concentrated in a small region called the nucleus, the proposed his model based on what evidence was available at that time on properties of atoms. However, they should be able to explain the basic properties of matter.įor example, when the electron was first discovered, then JJ Thompson proposed the plum cake model in which he took into consideration the two main points -ġ) From the CRT experiments, it was clear that all matter had those electrons within then thus it would be logical to suppose that electrons were a part of atoms.Ģ) In order to explain the charge neutrality, he supposed that the sphere in which electrons are embedded is positively charged. Such models would be purely theoretical constructs. 1800 -Dalton proposed a modern atomic model based on experimentation not on pure reason. 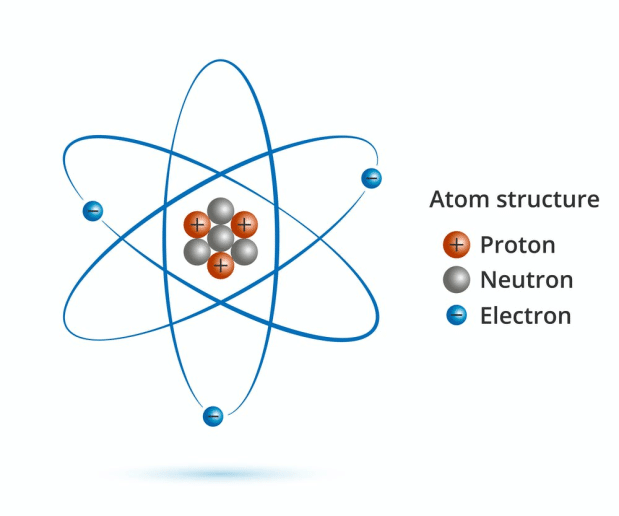
When the exact structure of the atom remained quite unknown, models were proposed based on experimental evidence of the properties of matter. An atomic model is a theory trying to explain the structure of the atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
